Evaluation effect of asthma on dentoalveolar morphology among children group
Abstract
Aim: The aim of this study was to evaluate the effect of asthma on dentoalveolar morphology in the developing child.
Methods: This study was conducted on 50 asthmatic Egyptian children and 50 non-asthmatic Egyptian children. Each child’s dental casts of the maxilla and mandible were analyzed. The upper and lower intercanine width, the upper and lower inter-molar width (UIMW-LIMW), the upper and lower arch length, and the palatal depth were determined.
Results: Children with asthma demonstrated higher mean values in more than one of the parameters measured when compared with the mean values of non-asthmatic children for both genders. In general, the UIMW of asthmatic children had lower mean values when compared with the mean value of non-asthmatic children, except when compared regarding gender. A Student t-test noted a significant difference in UIMW and LIMW in male children.
Conclusion: Asthma has an effect on upper and lower dental arch dimensions in both genders.
Keywords
Introduction
Asthma comprises a range of heterogeneous phenotypes that differ in presentation, etiology, and pathophysiology.[1] It has been documented that house dust mite-induced allergy and tobacco smoke allergy have consistently been found to trigger an exacerbation of asthma in children. Also, in children with a strong family history of asthma and fetuses exposed to maternal smoking during pregnancy, have a fourfold increase in the risk of developing “wheezing illnesses”.[2,3]
Epidemiological studies have suggested that certain types of outdoor air pollution (a low sulfur and a high diesel particulate environment) increased the severity of childhood asthma. Short-term fluctuations in the levels of air pollution may provoke emergency admissions for asthma or may aggravate existing chronic asthma. Other factors can act as triggers in exacerbating asthma in children, such as viral infections, exercise, changes in weather patterns, cold air, and psychological factors.[4]
The clinical onset of an asthmatic episode may occur over minutes (acute) or hours and days. Therefore, asthma can present in four gradients of severity depending on the evolution of the asthma attack.[5] Asthma is a chronic inflammatory disease that causes the airways to narrow and produce additional mucus which leads to difficulty breathing. It is characterized by the obstruction of the airflow for a period. This condition is either reversible, either spontaneously or controlled with the support of medication.[6]
Mouth breathing can play a crucial role in the growth and the development of the oral cavity and nasal cavity. In mouth breathing, the tongue position within the oral cavity is depressed, and the balance between the forces from the tongue and cheeks is different when compared with healthy children.[7]
The following three contacts play an important role in growth and development of the oral and nasal cavity. First, a competent lip seal; second, the contacts between the tip of the tongue and the lingual surfaces of the upper central incisors; and third, the contact of the soft palate with the tongue base. When the competent lip seal cannot be achieved, the tongue posture is on the floor of the oral cavity (depression of the tongue). The expansive lateral forces of the tongue against the palate are lost, and there are unopposed medial forces of the buccinators and the masseter muscles.[8] The effect is additionally effected by a pressure differential across the hard palate in the absence of nasal airflow, leading to a narrow and high hard palate.[9]
The discrepancy between the buccinator mechanism and the tongue can move the teeth, leading to dental malocclusion.[10,11] According to Moss’ theory of the Functional Matrix, bone growth dynamically responds to both function and the attached soft tissue forces.[12] This disturbance in the oral function results in abnormal growth, and the development of soft tissue and bony structures can cause the craniofacial complex to morph. The dentoalveolar morphology can change the dental arch width, dental arch length, and PD.[13,14]
Based on previous reports, this study focused on the evaluation of changes in the dentoalveolar morphology in asthmatic children.
Methods
Subjects and selection
This study was conducted on 100 Egyptian children of either gender. The age group selected for this study was 7-12 years old; this is because the first permanent molar erupts at 6 years old in a majority of children. The study excluded children more than 12 years old. The majority of children in this age group have 1 or more unerupted canines.
The study group consisted of 50 asthmatic children with mild or moderate persistent asthma that were selected from the ACIRU, Mansoura University, Children’s Hospital. The control group consisted of 50 non-asthmatic children, who were recruited from the outpatient clinic of the Pediatric Dentistry Department, Faculty of Dentistry, Mansoura University. The non-asthmatic children were present in the clinic for routine dental checkups.
The children involved in this study had the following inclusion criteria: the child has had a diagnosis of asthma for at least four years; the child should not have had previous orthodontic treatment or any abnormal oral habits; children without any abnormal physical growth; and, children with class I Angle’s molar orthodontic classification and facial symmetry.
Children were excluded from this study for 1 or more of these causes: children or his/her parents refused to participate in the study; missing 1 or 2 deciduous, or permanent canines, or permanent first molars, or grossly destructive caries. Children with retrognathia or who require a rapid intervention by a physician’s unit were excluded. The inclusion and exclusion criteria were selected from a previous study,[14] and our additional criteria.
Rules of the Ethics Committee of the Faculty of Dentistry, Mansoura University and medical ethics in dealing with children and parents were followed. All requirements of infection control and sterilization were followed. The parents were informed in detail about the objective of this study, and their right to refuse or stop participating at any time. The parents signed the Arabic agreement to participate in the study.
Dental arch assessment
After an oral examination of the children, casts were prepared as follows: a suitable pediatric plastic impression tray was selected and filled with silicone rubber base impression material; the upper and lower dental arch impressions were made; the impressions were poured with dental stone and allowed to dry. Certain points were identified on the dental cast to measure the width and length,[14,15] with a modified electronic digital caliper [Figure 1]. All of the measurements were completed under similar conditions and recorded. These measurements were repeated three times at one-week intervals. The average of the three readings was recorded for each case in millimeters.
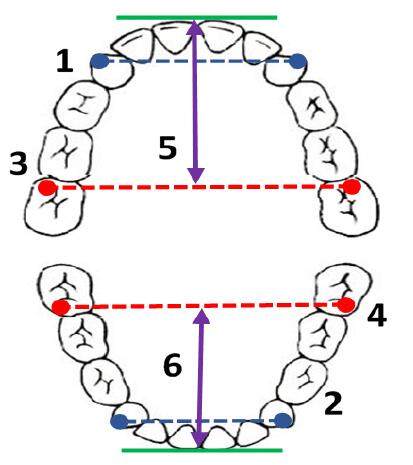
Figure 1. Measurement of dental cast. 1: Upper inter canine width; 2: lower inter canine width; 3: upper intermolar width; 4: lower inter molar width; 5: upper arch length; 6: lower arch length
Inter-canine width
The horizontal distance between the cusps of the upper canines was measured to obtain the upper intercanine width (UICW), and in the same manner, the distance between the cusps of the lower canines was measured to obtain the lower intercanine width (LICW) for each child.
Inter-molar width
The horizontal distance between the mesiobuccal cusps of the right and left maxillary first molars was measured to determine the upper inter-molar width (UIMW) and similarly the distance between the mandibular right and left first molars in the lower arch (LIMW) was recorded for each child.
Arch length
The horizontal distance from the inter-molar width line to the labial surface of the central incisors was used to obtain the upper arch length (UAL) and the lower arch length (LAL).
Palatal depth
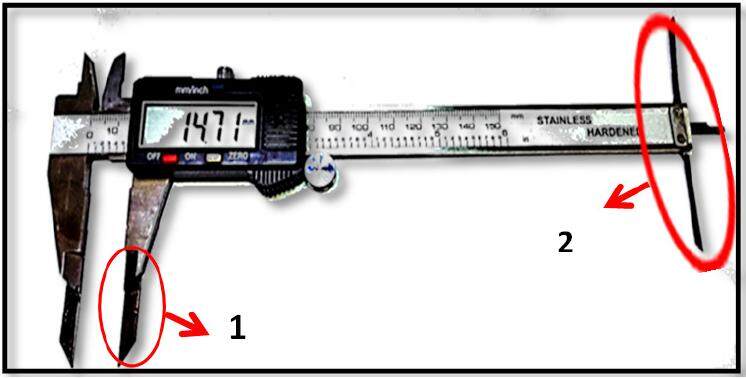
The palatal depth (PD) was measured vertically from the inter-molar width line to the palatal vault. At the suggestion of Qasem FA, a modification of the electronic digital caliper was made to a conventional electronic digital caliper (Shenzhen Cycwiss Technology Co., Ltd, China) [Figure 2]. This modification was completed with the help of an engineer in the field of sensitive measurement devices. Components were added to the caliper that facilitated the measurements (added 4 cm to the jaw length and 9 cm for the T-shape).
Statistical analysis
Data were tabulated, coded, and analyzed using the computer program IBM SPSS software package version 20.0. Quantitative data were described using range (minimum and maximum), mean, and standard deviation. The significance of the obtained results was judged at the 5% level. The Student t-test was used for normally distributed quantitative variables.
For standardization, a modified electronic digital caliper, and one examiner (Qasem FA) collected all of the data. The measurements were completed under the same conditions and were recorded 3 times at 1-week intervals for each measurement. The average of the 3 readings was recorded for each measurement in millimeters.
Results
One hundred Egyptian children of both genders between the age of 7-12 years were enlisted into the study [Table 1]. According to the Guidelines for Evaluating and Expressing the Uncertainty of NIST Measurement Results,[16] the accuracy of measurements by a modified electronic digital caliper for dental casts was in the range of 0.02 to 0.05 mm. The range of percentage of accuracy calculated for all measurements were 95.93-98.85%.
Comparison between the two groups according to demographic data
| Group | Asthmatic (n = 50) | Non-asthmatic (n = 50) | Total (n = 100) |
|---|---|---|---|
| Gender, n (%) | |||
| Male | 30 (60) | 22 (44) | 52 (52) |
| Female | 20 (40) | 28 (56) | 48 (48) |
| Age (years) | |||
| Min-Max | 7-12 | 7-12 | 7-12 |
| Mean ± SD | 8.73 ± 1.29 | 8.83 ± 1.35 | 8.78 ± 1.32 |
The mean value of the UICW in the non-asthmatic group was 32.19 ± 2.18 mm, while the mean value in the asthmatic group was 32.66 ± 3.02 mm. Statistical analysis of the UIMW was statistically insignificant (P = 0.379). The lower mean value in the non-asthmatic group was 49.06 ± 2.06 mm when compared with the asthmatic group mean value which was 49.91 ± 3.01 mm and is statistically insignificant (P = 0.102). In the UAL, the lower mean value in the non-asthmatic group was 30.09 ± 2.38 mm when compared with the asthmatic group which was 30.50 ± 2.72 mm. Statistical analysis of the UAL was statistically insignificant (P = 0.418). A higher mean value of the PD in the asthmatic group was 14.12 ± 1.65 mm when compared with non-asthmatic group which was 13.94 ± 1.20 mm. Statistical analysis of the PD was statistically insignificant (P= 0.516). Also, compared between asthmatic and non-asthmatic depend on the gender as present in tables [Tables 2 and 3].
Comparison between the two groups according to upper arch dimensions in male children
| Group | Asthmatic (n = 30) | Non-asthmatic (n = 22) | t | P |
|---|---|---|---|---|
| Upper inter canine width | ||||
| Min-Max (mm) | 25.72-38.50 | 28.91-36.92 | 0.42 | 0.67 |
| Mean ± SD (mm) | 32.84 ± 3.40 | 32.51 ± 2.21 | ||
| Upper inter molar width | ||||
| Min-Max (mm) | 45.34-54.93 | 44.98-53.62 | 2.89 | 0.00 |
| Mean ± SD (mm) | 50.58 ± 2.94 | 48.59 ± 2.0 | ||
| Upper arch length | ||||
| Min-Max (mm) | 21.84-34.86 | 26.41-35.0 | 0.60 | 0.54 |
| Mean ± SD (mm) | 30.66 ± 3.14 | 30.18 ± 2.27 | ||
| Palatal depth | ||||
| Min-Max (mm) | 10.48-17.58 | 11.65-15.98 | 0.75 | 0.45 |
| Mean ± SD (mm) | 14.17 ± 1.81 | 13.84 ± 1.10 |
Comparison between the two groups according to upper arch dimensions in female children
| Group | Asthmatic (n = 20) | Non-asthmatic (n = 28) | t | P |
|---|---|---|---|---|
| Upper inter canine width | ||||
| Min-Max (mm) | 28.26-36.59 | 27.78-36.0 | 0.67 | 0.50 |
| Mean ± SD (mm) | 32.39 ± 2.38 | 31.94 ± 2.16 | ||
| Upper inter molar width | ||||
| Min-Max (mm) | 42.29-52.99 | 45.76-53.22 | 0.72 | 0.47 |
| Mean ± SD (mm) | 48.91 ± 2.90 | 49.43 ± 2.06 | ||
| Upper arch length | ||||
| Min-Max (mm) | 25.49-35.88 | 25.43-36.0 | 0.38 | 0.70 |
| Mean ± SD (mm) | 30.27 ± 1.97 | 30.02 ± 2.51 | ||
| Palatal depth | ||||
| Min-Max (mm) | 12.25-17.34 | 12.0-16.92 | 0.10 | 0.91 |
| Mean ± SD (mm) | 14.05 ± 1.41 | 14.01 ± 1.28 |
In the lower arch, the higher mean value of the LICW in the asthmatic group was 26.50 ± 3.10 mm compared with the non-asthmatic group which was 26.22 ± 2.44 mm. Statistical analysis of the LICW was statistically insignificant (P = 0.613). A higher mean value of the LIMW in the asthmatic group was 44.31 ± 3.57 mm when compared with the non-asthmatic group which was 42.94 ± 2.87 mm. Statistical analysis of the LIMW was statistically significant (P = 0.038). A lower mean value of the LAL in the non-asthmatic group was 26.44 ± 1.93 mm when compared with the asthmatic group which was 27.71 ± 2.49 mm. Statistical analysis of the LAL was statistically significant (P = 0.005). In addition, compared between asthmatic and non-asthmatic depend on the gender as present in tables [Tables 4 and 5].
Comparison between the study and control group according to lower arch dimensions in male children
| Group | Asthmatic (n = 30) | Non-asthmatic (n = 22) | t | P |
|---|---|---|---|---|
| Lower inter canine width | ||||
| Min-Max (mm) | 21.13-36.89 | 23.76-36.89 | 0.60 | 0.54 |
| Mean ± SD (mm) | 26.50 ± 3.16 | 27.0 ± 2.60 | ||
| Lower inter molar width | ||||
| Min-Max | 40.03-52.99 | 38.68-52.99 | 2.10 | 0.04 |
| Mean ± SD (mm) | 44.70 ± 3.37 | 42.80 ± 3.02 | ||
| Lower arch length | ||||
| Min-Max (mm) | 22.55-31.95 | 23.92-31.95 | 1.31 | 0.19 |
| Mean ± SD (mm) | 27.48 ± 2.10 | 26.75 ± 1.83 |
Comparison between the study and control group according to lower arch dimensions in female children
| Group | Asthmatic (n = 20) | Non-asthmatic (n = 28) | t | P |
|---|---|---|---|---|
| Lower inter canine width | ||||
| Min-Max (mm) | 22.75-35.78 | 21.87-30.54 | 1.18 | 0.24 |
| Mean ± SD (mm) | 26.50 ± 3.09 | 25.60 ± 2.16 | ||
| Lower inter molar width | ||||
| Min-Max (mm) | 37.80-52.48 | 38.98-52.43 | 0.67 | 0.50 |
| Mean ± SD (mm) | 43.71 ± 3.87 | 43.05 ± 2.80 | ||
| Lower arch length | ||||
| Min-Max (mm) | 23.44-35.06 | 21.10-29.96 | 2.60 | 0.01 |
| Mean ± SD (mm) | 28.07 ± 3.0 | 26.19 ± 1.99 |
Discussion
The effects of asthma on general body growth and development of children can manifest itself in different ways. Asthma can effect functionally (mechanically) the development of bone structures and soft tissue by changing the mode of breathing to mouth breathing. This disturbance in the development of the craniofacial skeleton depends on the capsular matrix theory of growth.[17]
The present study demonstrated a higher mean value in the UICW for the asthmatic children compared with the non-asthmatic children which are statistically insignificant. On the contrary, the higher value observed in both asthmatic and non-asthmatic male children when compared with asthmatic and non-asthmatic female children was statistically insignificant. Most of the previous studies[13,15,18] did not measure the UICW of the asthmatic children. However, the result of the present study of the UICW when compared with the non-asthmatic children according to gender agreed with the work of Al-Khafaji.[19] Al-Khafaji[19] found an increased mean value of the UICW in normal male children, which agrees with the work of Kumar and Nandlal.[18] Kumar and Nandlal[18] noted a higher mean value of the UICW when comparing asthmatic and non-asthmatic children in both genders. The UIMW demonstrated a higher value of asthmatic children than non-asthmatic children without a statistically significant difference. The result of the UIMW of this study agrees with Kumar and Nandlal[18] work which suggested that the inter-molar width had a higher mean value of the asthmatic children than the non-asthmatic children in both genders without significant difference.
Lione et al.,[15] Malhotra et al.[20] and Elisabet and Wenzel[21] disagree with the results of Nandlal’s study. They established that the UIMW in asthmatic children had a lower mean value when compared with the non-asthmatic children in both genders. This study disagrees with Yavuz and Oktay[22] who found an increased mean value of the UIMW in normal male children. The difference in the results of our study may be due to the difference in methodology to evaluate the arch dimensions. Yavuz and Oktay[22] depended on a computerized program, a 3D scan, or they used different landmarks when measuring points on the dental cast.
The UAL had a higher value in asthmatic children when compared with non-asthmatic children; however, this is statistically insignificant. In addition, a higher mean value presented in asthmatic and non-asthmatic male children than asthmatic and non-asthmatic female children without a statistically significant difference.
The result of the UAL in this study agrees with previous studies.[13,18] The previous studies found that the UAL in asthmatic children had a higher mean value than non-asthmatic children in both genders. Höjensgaard and Wenzel[21] and Gungor and Turkkahraman[23] disagree with the results of this study. They concluded that the UAL had a lower mean value in asthmatic children than in non-asthmatic children. Zafarmand et al.[24] found a lower mean value of the UAL in normal male children when compared with non-asthmatic children according to gender. The difference may be due to asthmatic children who participated in this study had mild and moderate persistent asthma controlled with medication that allows dental arch changes as though they are healthy children. Other papers do not mention the severity of asthma and if children’s asthma is controlled.
The PD had a higher mean value in asthmatic children compared with non-asthmatic children without identifying a statistically significant difference. The higher value of the PD in our present study agrees with previous studies which have reported that asthmatic children had a higher mean value. This value is statistically insignificant when compared with the same age group of non-asthmatic children.[13,18]
Lione et al.[15] and Höjensgaard and Wenzel[21] found a highly significant difference in the PD in asthmatic children. In addition, Abdulmawjood et al.[25] found a decreased mean value of the PD in normal male children when compared with non-asthmatic children according to gender. The possible explanation of the higher mean value in the PD of the studies[15,21] may be due to the effect of mouth breathing in some the cases of asthmatic children and insufficient control of their asthma by medication.
The present study observed a statistically insignificant difference in the LICW in asthmatic children compared with non-asthmatic children. Non-asthmatic children demonstrated a higher mean value in males which is statistically significant. Most of the previous studies[13,15,18,21] did not evaluate the relationship between asthma in children and the mandibular dental arch. However, the result of the present study of the LICW when compared with non-asthmatic children according to gender agrees with Lara-Carrillo et al.[26] Lara-Carrillo et al.[26] found an increased mean value of the LICW in normal male children, and Kumar and Nandlal[18] found a higher mean value of lower interincisal width between asthmatic and non-asthmatic children for both genders.
The result of the LIMW demonstrated a statistically significant difference between the asthmatic children compared with the non-asthmatic children (except in male children). In addition, non-asthmatic females had a higher mean value in the LIMW when compared to non-asthmatic males without statistical significance. This result is in accordance with the previous study,[18] of Höjensgaard and Wenzel[21] who suggested that the inter-molar width had a higher value in asthmatic children in both genders without a statistically significant difference. Malhotra et al.[20] disagreed with the result of the LIMW in our study. They reported that the LIMW in asthmatic children had a lesser mean value than non-asthmatic children in both genders, Al-Zubair[27] suggested that male non-asthmatic children had a higher mean value when compared with non-asthmatic females. The variance in the results may be due to the asthmatic children included in this study had mild and moderate asthma; whereas, the previous studies did not delineate the diagnostic levels of asthma.
The LAL displayed a statistically significant difference in asthmatic children when compared with non-asthmatic children. The LAL result of this study agreed with a previous study which demonstrated a statistically significant difference in the LAL of asthmatic children when compared with non-asthmatic children.[18] On the other hand, Kumar and Nandlal[13] disagreed with the result of this study. Kumar and Nandlal found that asthmatic children had a lower mean value when compared with non-asthmatic children of both genders. The difference with the other studies may be attributed to different ethnic groups, sample sizes, and environmental factors.
Even though our research has accomplished its aims, there were some unavoidable limitations. First, because of time limitations our research was conducted only on a small number of asthmatic and non-asthmatic children. Second, the study was conducted on two types of severity classifications of asthma (mild and moderate). Therefore, the study should have involved different age groups with a larger number of children, dependent on gender, and using different severity classifications of asthma.
This article focused on evaluate effect of asthma on dentoalveolar morphology unstudied enough and previous studies did not focus on asthmatic children in particular but examined attendant variables with asthma.
Asthma effects the development of the dental arches and can cause malocclusion. Therefore, it is important to make an accurate diagnosis and an efficienct treatment plan to prevent exaggeration of oral cavity problems in asthmatic children.
Authors’ contributions
Manuscript’s preparation, design and literature search: F.A. Qasem
Manuscript’s review: S.M. Awad, T. El-Desoky, H.M. Shalan
Acknowledgments
We thank Dr. Jon Wagner for his contribution in language editing and Dr. Salwa Hegazy for her support in statistic checking.
Financial support and sponsorship
None.
Conflicts of interest
There are no conflicts of interest.
Patient consent
Obtained.
Ethics approval
The procedures followed were in accordance with the rules of the Ethics Committee of the Faculty of Dentistry, Mansoura University and Medical Ethics in dealing with children and parents. All steps of infection control and the sterilization protocol were followed.
REFERENCES
1. Dezateux C, Stocks J, Dundas I, Fletcher ME. Impaired airway function and wheezing in infancy: the influence of maternal smoking and a genetic predisposition to asthma. Am J Respir Crit Care Med 1999;159:403-10.
2. Subbarao P, Mandhane PJ, Sears MR. Asthma epidemiology, etiology and risk factors. CMAJ 2009;181:E181-90.
5. Weddell JA, Sanders BJ, Jones JE. Dental Problems of Children with Special Health Care Needs. In: Dean JA. Dentistry for the Child and Adolescent. 9th ed. Mosby: Elsevier; 2011:460-86.
6. Thomas MS, Parolia A, Kinabala M, Vikram M. Asthma and oral health: a review. Aust Dent J 2010;55:128-33.
7. Solow B, Siersbaek-Nielsen S, Greve E. Airway adequacy, head posture, and craniofacial morphology. Am J Orthod 1984;86:214-23.
8. Yawn BP. Factors accounting for asthma variability, achieving optimal symptom control for individual patients. Prim Care Respir J 2008;17:138-47.
9. Principato JJ. Upper airway obstruction and craniofacial morphology. Otolaryngol Head Neck Surg 1991;104:881-90.
10. Martínez EJL, Oma-a VE. Dental malocclusion and bony abnormalities in girls with nasopharyngeal obstruction of allergic origin. Pract Odontol 1988;9:8-12.
12. Moss ML, Salentijn L. The primary role of functional matrices in facial growth. Am J Orthod 1969;55:566-77.
13. Kumar SS. Nandlal B. Evaluation of changes in the dentoalveolar morphology in children with asthma. Pediatric Dent J 2012;22:95-102.
14. Klein JC. Nasal respiratory function and craniofacial growth. Arch Otolaryngol Head Neck Surg 1986;112:843-9.
15. Lione R, Buongiorno M, Franchi L, Cozza P. Evaluation of maxillary arch dimensions and palatal morphology in mouth-breathing children by using digital dental casts. Int J Pediatr Otorhinolaryngol 2014;78:91-5.
16. Taylor BN, Kuyatt CE. Guidelines for Evaluating and Expressing the Uncertainty of NIST Measurement Results. Available from: https://www.nist.gov/sites/default/files/documents/pml/pubs/tn1297/tn1297s.pdf. [Last accessed on 28 Mar 2017].
17. Freitas FCN, Bastos EP, Primo LS, de Freitas VL. Evaluation of the palatal dimensions of patients with perennial allergic rhinitis. Int J Paediatr Dent 2001;11:365-71.
18. Kumar SS, Nandlal B. Effects of asthma and inhalation corticosteroids on the dental arch morphology in children. J Indian Soc Pedod Prev Dent 2012;30:242-9.
19. Al-Khafaji TJ. Dental arch dimensions of patients with class III malocclusion of Iraqi sample. Med J Babylon 2011;8:33-48.
20. Malhotra S, Gupta V, Pandey RK, Singh SK, Nagar A. Dental consequences of mouth breathing in the pediatric age group. Int J Oral Health Sci 2013;3:79-83.
21. Höjensgaard E, Wenzel A. Dentoalveolar morphology in children with asthma and perennial rhinitis. Eur J Orthod 1987;9:265-70.
22. Yavuz I, Oktay H. Changes in the dental arches that occurred in transition from mixed dentitions to permanent dentitions: a longitudinal study. J Dent Fac Atatürk Univ 2006;16:8-13.
23. Gungor AY, Turkkahraman H. Effects of airway problems on maxillary growth: a review. Eur J Dent 2009;3:250-4.
24. Zafarmand AH, Mina M, Zafarmand M. Maxillary arch dimension changes of 3-5 years old Filipino children. Nov In Biomed 2014;2:126-30.
25. Abdulmawjood AA, Ahmed MK, Al-Saleem Ne'am R. Palatal depth and arch parameter in class I open bite, deep bite and normal occlusion. Iraqi Orthod J 2005;1:26-31.
26. Lara-Carrillo E, González-Pérez JC, Kubodera-Ito T, Montiel-Bastida NM, Esquivel-Pereyra GI. Dental arch morphology of mazahua and mestizo teenagers from central Mexico. Braz J Oral Sci 2009;8:92-6.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Awad SM, El-Desoky T, Shalan HM, Qasem FA. Evaluation effect of asthma on dentoalveolar morphology among children group. Stomatological Dis Sci 2017;1:22-8. http://dx.doi.org/10.20517/2573-0002.2016.14
AMA Style
Awad SM, El-Desoky T, Shalan HM, Qasem FA. Evaluation effect of asthma on dentoalveolar morphology among children group. Stomatological Disease and Science. 2017; 1: 22-8. http://dx.doi.org/10.20517/2573-0002.2016.14
Chicago/Turabian Style
Awad, Salwa Mohamed, Tarek El-Desoky, Hanaa Mahmoud Shalan, Fathi Abdulraqep Qasem. 2017. "Evaluation effect of asthma on dentoalveolar morphology among children group" Stomatological Disease and Science. 1: 22-8. http://dx.doi.org/10.20517/2573-0002.2016.14
ACS Style
Awad, SM.; El-Desoky T.; Shalan HM.; Qasem FA. Evaluation effect of asthma on dentoalveolar morphology among children group. Stomatological. Dis. Sci. 2017, 1, 22-8. http://dx.doi.org/10.20517/2573-0002.2016.14
About This Article
Copyright
Author Biographies


Data & Comments
Data
 Cite This Article 8 clicks
Cite This Article 8 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.